Phosphorothioate Oligos
Phosphorothioate
Phosphorothioate oligonucleotides (Phosphorothioate, PTO)
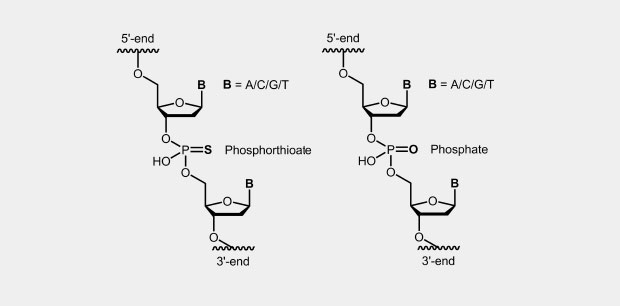
The phosphorothioate modification of the DNA backbone is one of the first and still widely used modification for antisense oligonucleotides (antisense oligos of the first generation). In the phosphate backbone, one of the two oxygen atoms which is not involved in the internucleotide bridge, is replaced by a sulfur atom (Fig.). Due to the four different substituents, the central phosphorus atom is a chiral center which effects diastereomer formation.
In oligonucleotides, all (Full-PTO) or just single phosphates (Part-PTO) can be replaced by phosphorothioate. Often it seems to be sufficient to incorporate two to four PTOs at the end of the oligos to achieve the desired stability against degradation.
|
|
Order information:
Phosphorothioates are presented in the order system of biomers.net by asterisks (*). If all or just single phosphodiester should be replaced by phosphorothioates, a * have to be inserted into the sequence at the respective position.
Examples:
- A normal sequence without phosphorothioates:
agcgtgacgtggacgtgagtga - The identical sequence, but as a "full-PTO-oligo" (all inter-nucleotidic phosphodiester were changed to PTO):
a * g * c * t * g * g * a * c * t * g * g * g * a * c * t * g * g * a * g * t * g * a - The sequence with single PTOs, in this case three PTO at both ends:
a * g * c * gtgacgtggacgtgag * t * g * a - The sequence with a PTO-bridge at the 3 'end:
agcgtgacgtggacgtgagtg * a
If a PTO-Oligo should be modified at one end, for example with a fluorescent dye, it is often useful, to "stabilize" also the phosphate bridge between the oligo and the modification (synthesizing as a PTO).
At a 5'-modification, in this case a * is also added to the 5'-end of the oligos (at a 3'-modification according to the 3'-end) and the desired modification is selected in the menu:
- * a * g * c * t * g * g * a * c * t * g * g * g * a * c * t * g * g * a * g * t * g * a, 5'-modification: biotin: PTO between biotin and oligo
Literature:
- An antisense oligonucleotide primer. Hogrefe RI; Antisense and Nucleic Acid Drug Development (1999), 9(4): 351-357.